High Dose Vitamin C IV Therapy for Cancer Patients
If you’re going through chemotherapy, radiotherapy or immunotherapy, vitamin C IV therapy can help your body cope — reducing side effects, supporting your immune system and helping you feel stronger through treatment.
- Support your immune system during treatment
- Help reduce chemotherapy side effects
- Feel stronger between treatment cycles



“Many of our patients come to us feeling exhausted by their treatment. What vitamin C IV therapy does is give the body the support it needs to keep going — helping patients feel stronger, recover faster between cycles, and tolerate their treatment better. The research is encouraging, and we see the difference it makes every day.”
- Dr Saskia Kloppenburg Vieth, Consultant in Integrative Oncology

GMC-Registered Doctors
Research-Backed Protocols
Tailored to Your Diagnosis
Trusted by Hundreds of Patients

Vitamin C IV Therapy — Designed for Cancer Patients
At Wellbeing Medical Group, we use high dose vitamin C IV therapy specifically to support people going through cancer treatment. This is not a general wellness drip. Every protocol we design is based on your diagnosis, your therapy plan and your individual needs.
We work alongside your NHS oncologist, not instead of them. When you bring us your reports and scan results, Dr Saskia reviews them personally and builds an IV therapy plan that complements what your hospital team is already doing.
Because our clinic focuses entirely on cancer care, we understand how vitamin C interacts with different types of chemotherapy, radiotherapy and immunotherapy. That means we know when it can help, how to dose it safely, and what to look out for along the way.
Our IV infusion team specialises in cancer care. Every member of our clinical team understands the unique needs of patients going through treatment, and we stay up to date with the latest research so that your protocol reflects current best practice.
Our clinic on Tower Bridge Road is easily accessible by public transport and within walking distance of London Bridge station. We’ve designed the space to feel calm and private — a long way from a hospital waiting room.
From the moment you arrive, our team is here to look after you. The clinic is a quiet, comfortable space where you can relax during your infusion. Many of our patients tell us it’s the most restful part of their week.
We offer flexible scheduling around your hospital treatment calendar. Booking is straightforward and our team will help you find times that work with your existing appointments.
We begin with a free consultation to understand your diagnosis and treatment history. Dr Saskia and the medical team will then design a protocol tailored to your specific case and provide you with clear pricing. There is no obligation to proceed — this step simply ensures any recommendation we make is right for you.
What Is Vitamin C IV Therapy?
Vitamin C IV therapy delivers high doses of vitamin C directly into your bloodstream through a drip. When taken by mouth, even at high doses, your body tightly controls how much reaches your blood — plasma levels quickly plateau. An IV drip bypasses these limits entirely, achieving concentrations up to 70 times higher than oral supplements.
At these higher concentrations, vitamin C works differently than it does at everyday levels. Rather than acting as a simple antioxidant, it creates a pro-oxidant effect that can selectively target cancer cells while leaving healthy cells largely unaffected.
Research has shown that high dose vitamin C IV therapy can support immune function, and in certain cancer types may help sensitise tumours to chemotherapy and radiotherapy. Clinical studies have also found it can improve quality of life for patients going through treatment.
This is not a one-off treatment. The benefits of vitamin C IV therapy build over time through repeated sessions, which is why Dr Saskia designs each patient’s protocol as a course of infusions rather than a single appointment.

Our Patients - Empowering People Lives
Fiona
Diagnosis: Adenocarcinoma non-small cell lung cancer
After one cycle of chemotherapy and immunotherapy, Fiona decided the side effects were too much. She came to us looking for a different way to support her body alongside her hospital care.
Dr Saskia reviewed her biopsy results, designed an integrative plan around her needs, and worked with her throughout.
Her oncologist monitored her progress closely — and Fiona was able to maintain her quality of life and disease stability.
Graeme
Diagnosis: Adenocarcinoma Oesophagus
Graeme contacted us before he’d even had his final diagnosis confirmed.
He wanted to be prepared — to have a plan that worked alongside whatever his NHS team recommended.
Dr Saskia built an integrative care plan around his surgery and treatment, focused on strengthening his body and managing side effects.
Isis
Diagnosis: Hodgkin Lymphoma
Isis was facing six months of chemotherapy for an aggressive diagnosis.
She came to us wanting to do everything she could to reduce side effects and give the chemotherapy the best chance of working.
Dr Saskia designed an intensive protocol alongside her hospital treatment, and Isis committed fully — attending every session and immersing herself in the research behind her care.
Mon – Fri 9:00am – 6:00pm

Mon – Fri 9:00am – 6:00pm

- 185 Tower Bridge Road, London
Vitamin C IV Therapy — Common Questions
Vitamin C IV therapy has a strong safety profile and is generally very well tolerated by cancer patients. Side effects are uncommon and usually mild. Clinical research consistently shows that high dose vitamin C delivered intravenously is safe when administered under proper medical supervision.
That said, there are a small number of situations where vitamin C IV therapy may not be appropriate — for example, certain kidney conditions or specific drug interactions. Before starting any treatment, Dr Saskia and the medical team review your full diagnosis, your hospital treatment plan and your medical history to make sure it’s right for you. If it isn’t, we will tell you clearly before you commit to anything.
Because we work alongside your NHS oncologist, everyone involved in your care knows what you’re receiving. This joined-up approach helps ensure your safety throughout.
When vitamin C is delivered in high doses directly into the bloodstream, it behaves very differently from the vitamin C you get from food or supplements. At these concentrations, it acts as a pro-oxidant — creating a reaction that can selectively target cancer cells while leaving healthy cells largely unaffected.
Research has also shown that high dose vitamin C can support your immune system by boosting the activity of key immune cells, including T-cells and natural killer cells. This is particularly important during chemotherapy, when your immune defences are under strain.
Additionally, clinical trials in certain cancer types suggest that vitamin C may help sensitise tumour cells to chemotherapy and immunotherapy, potentially improving how well your hospital treatment works. The benefits are cumulative — they build over a course of infusions rather than a single session, which is why Dr Saskia designs every protocol as a planned series of treatments.
Yes. This is one of the most well-supported benefits of vitamin C IV therapy in cancer care. Multiple clinical studies have found that patients receiving high dose vitamin C alongside their treatment report less pain, less fatigue and a better overall sense of wellbeing.
For patients in palliative care or those whose hospital treatment has ended, the evidence is particularly encouraging. Research has shown meaningful improvements in quality of life, pain management and physical function. One study found that patients with advanced cancer who received vitamin C IV therapy had significantly better outcomes in terms of both comfort and survival time compared to those who did not.
Many of our patients tell us that their vitamin C sessions are one of the things that helps them feel most like themselves during a very difficult time. It’s not a cure — we would never claim that — but the difference it can make to how you feel day to day is real and well documented.
We are not a general IV drip clinic. Wellbeing Medical Group exists specifically to support cancer patients, and every aspect of our vitamin C IV therapy is designed around that.
Your protocol is designed by Dr Saskia, who reviews your hospital reports, understands your diagnosis and builds a plan that works alongside your NHS treatment. Our nursing team specialises in cancer care — they understand what you’re going through and how to support you through each session.
We are CQC regulated, which means we meet the same clinical standards as NHS services. And if you can’t travel to our London clinic, our nursing team can deliver your vitamin C IV therapy at home, anywhere in the UK.
If you’re looking for vitamin C IV therapy from a team that understands cancer — not just drips — call us on 020 3376 4835 or book a free consultation.
Why Patients Choose Vitamin C IV Therapy
Patients come to us for vitamin C IV therapy at different stages of their cancer journey — some before treatment starts, some during chemotherapy, and some after hospital treatment has ended. The reasons vary, but the most common are:
To reduce treatment side effects. Many patients find that vitamin C IV therapy helps manage the fatigue, nausea and immune suppression that come with chemotherapy and radiotherapy. When your body has the support it needs, you’re better placed to complete your therapy course and recover between cycles.
For a fuller breakdown of how different IV protocols target specific side effects, see our guide to IV infusion therapy for managing cancer treatment side effects.
To support your immune system. Cancer treatment can suppress your immune system at the time you need it most. Vitamin C plays a critical role in immune function, supporting the cells your body relies on to fight infection and disease.
To complement your hospital treatment. Clinical trials in certain cancer types have shown that high dose vitamin C can sensitise tumour cells to chemotherapy and radiation, potentially improving treatment outcomes. Dr Saskia designs every protocol to work alongside what your NHS team is doing, not against it.
To feel stronger and more in control. A cancer diagnosis can leave you feeling powerless. Many of our patients tell us that having an active role in their care — doing something positive alongside their hospital treatment — makes a real difference to how they cope, both physically and mentally.
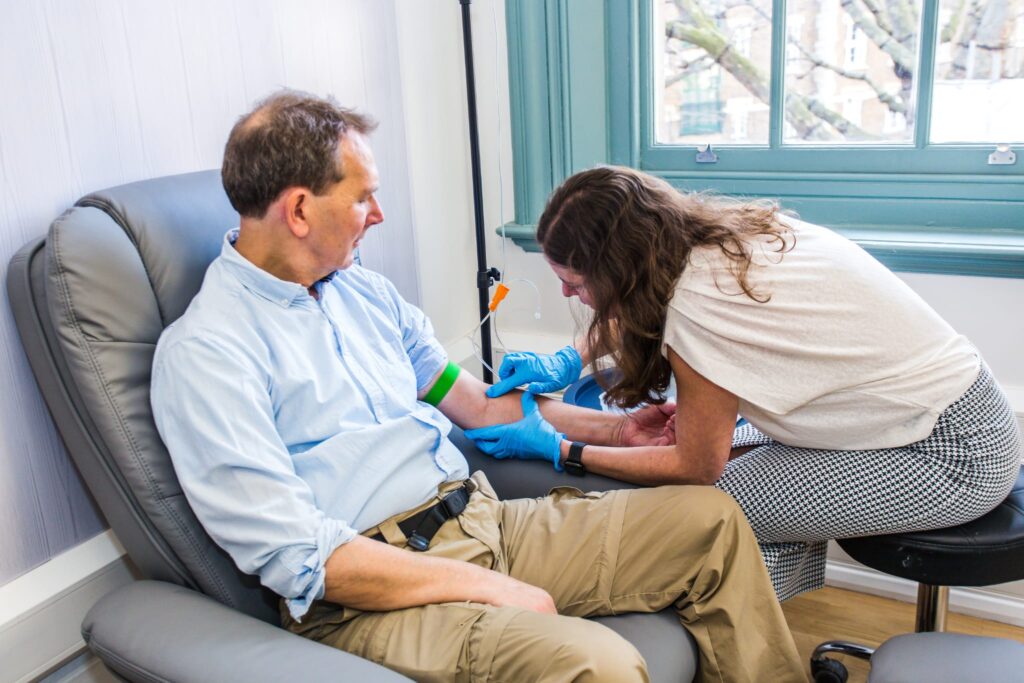
How Does Vitamin C IV Therapy Work?
When vitamin C is delivered in high doses directly into the bloodstream, it works in several ways to support cancer patients. Here’s what the research tells us, in plain terms:
At high concentrations, vitamin C creates a chemical reaction in the area around a tumour. Cancer cells are less able to defend themselves against this than healthy cells, which means the vitamin C can cause damage to the cancer while leaving normal tissue largely unaffected.
Clinical trials in certain cancer types have shown that vitamin C IV therapy can make tumour cells more sensitive to chemotherapy and radiotherapy. By increasing the stress on cancer cells, it may enhance the effect of your hospital treatment and help reduce the risk of resistance developing.
Vitamin C is essential for the production and function of immune cells, including T-cells and natural killer cells. During cancer treatment, when your immune system is under strain, high dose vitamin C can help maintain the defences your body needs.
A single infusion won’t deliver the full benefit. Vitamin C IV therapy works cumulatively — maintaining elevated levels in the body over a course of sessions is what produces the strongest therapeutic effect. That’s why Dr Saskia designs every protocol as a planned series of infusions, not a one-off appointment.
Vitamin C IV Therapy at Home — Anywhere in the UK
Your therapy plan is still designed by Dr Saskia and overseen by our medical team. The only difference is where you sit during your infusion.
For patients who are too unwell to travel, or who simply prefer to be treated at home, this service means you can access the same specialist cancer support wherever you are.
- IV therapy delivered by our nurses at your home
- Nurses available across the UK
- Therapy plan designed by Dr Saskia, delivered locally
- Same protocol and medical oversight as our London clinic

The Benefits of Vitamin C IV Therapy for Cancer Patients
You might be wondering why vitamin C needs to be given through a drip rather than taken as a tablet. The doses needed for cancer support are far higher than your body can absorb through the digestive system.
Full Absorption
Oral vitamin C is limited by your body’s absorption mechanisms — at higher doses, plasma levels plateau no matter how much you take. A vitamin C IV drip bypasses these limits entirely, achieving blood concentrations up to 70 times higher than oral supplements and reaching the therapeutic levels needed in cancer care.
Tailored to Your Treatment
Our vitamin C IV therapy is designed to work alongside your chemotherapy, radiotherapy or immunotherapy. Dr Saskia schedules your infusions to complement your hospital treatment cycles for the best possible outcome.
Precise Dosing
Every patient’s protocol is different. IV delivery allows our team to control exactly how much vitamin C you receive, adjusting the dose based on your diagnosis, your therapy stage and how you’re responding.
Faster Support
IV delivery means the vitamin C reaches your system immediately. For patients who are fatigued, immunosuppressed or struggling with side effects, this faster route can make a meaningful difference to how quickly you feel the benefit.

Is Vitamin C IV Therapy Safe?
High dose vitamin C IV therapy has a strong safety profile and is generally very well tolerated by cancer patients. Most people experience no side effects at all.
Before we begin any treatment, our medical team carries out a thorough review of your diagnosis, your hospital treatment plan and your full medical history.
There are a small number of situations where vitamin C IV therapy may not be appropriate — and if that applies to you, we will tell you clearly before you commit to anything.
We also stay in close communication with your NHS oncologist throughout, so that everyone involved in your care knows what you’re receiving and can flag any concerns.
Why Choose Wellbeing Medical Group for Vitamin C IV Therapy?
We are not a general wellness clinic. Wellbeing Medical Group exists specifically to support cancer patients with integrative therapies that work alongside hospital treatment.
Every treatment plan is designed by Dr Saskia, a consultant in integrative oncology and one of three consultants at the Royal London Hospital for Integrated Medicine. She reviews your hospital reports, understands your diagnosis, and builds a vitamin C IV protocol that complements what your NHS team is already doing.
We are CQC regulated, which means we meet the same clinical standards as NHS services. Our nursing team is experienced in cancer care and available across the UK for home-based treatment.
If you’re looking for vitamin C IV therapy from a team that understands cancer — not just IV drips — we’d like to hear from you.

The following published studies inform the therapies and protocols described on this page.
Bryan Ngo et al, Targeting cancer vulnerabilities with high-dose vitamin C, Nat Rev Cancer 2019 May;19(5):271-282. doi: 10.1038/s41568-019-0135-7.
Polireddy K, Dong R, Reed G, Yu J, Chen P, Williamson S, et al. High dose parenteral Ascorbate inhibited pancreatic Cancer growth and metastasis: mechanisms and a phase I/IIa study. Sci Rep. 2017;7(1):17188.
Chen Q, Espey MG, Sun AY, Pooput C, Kirk KL, Krishna MC, et al. Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc Natl Acad Sci. 2008;105(32):11105–9.
Schoenfeld JD, Sibenaller ZA, Mapuskar KA, Wagner BA, Cramer Morales KL, Furqan M, et al. O 2 ·− and H 2 O 2 Mediated Disruption of Fe Metabolism Causes the Differential Susceptibility of NSCLC and GBM Cancer Cells to Pharmacological Ascorbate. Cancer Cell. 2017;31(4):487–500.e8.
Takahashi H, Mizuno H, Yanagisawa A. High dose intravenous vitamin C improves quality of life in cancer patients. Pers Med Universe. 2012;1(1):49–53.
Vollbracht C, Schneider B, Leendert V, Weiss G, Auerbach L, Beuth J. Intravenous vitamin C administration improves quality of life in breast cancer patients during chemo−/radiotherapy and aftercare: results of a retrospective, multicentre, epidemiological cohort study in Germany. In Vivo. 2011;25(6):983–90.
Yeom CH, Jung GC, Song KJ. Changes of terminal Cancer patients’ health‐related quality of life after high dose vitamin C administration. J Korean Med Sci. 2007;22(1):7.
Agathocleous M, Meacham CE, Burgess RJ, Piskounova E, Zhao Z, Crane GM, et al. Ascorbate regulates haematopoietic stem cell function and leukaemogenesis. Nature. 2017;549(7673):476–81.
Bonilla‐Porras AR, Jimenez‐Del‐Rio M, Velez‐Pardo C. Vitamin K3 and vitamin C alone or in combination induced apoptosis in leukemia cells by a similar oxidative stress signalling mechanism. Cancer Cell Int. 2011;11(1):19.
Cimmino L, Dolgalev I, Wang Y, Yoshimi A, Martin GH, Wang J, et al. Restoration of TET2 Function Blocks Aberrant Self‐Renewal and Leukemia Progression. Cell. 2017;170(6):1079–1095.e20.
Iamsawat S, Tian L, Daenthanasanmak A, Wu Y, Nguyen HD, Bastian D, et al. Vitamin C stabilizes CD81 iTregs and enhances their therapeutic potential in controlling murine GVHD and leukemia relapse. Blood Adv. 2019;3(24):4187–201.
Mingay M, Chaturvedi A, Bilenky M, Cao Q, Jackson L, Hui T, et al. Vitamin C‐induced epigenomic remodelling in IDH1 mutant acute myeloid leukaemia. Leukemia. 2018;32(1):11–20.
Aguilera O, Muñoz‐Sagastibelza M, Torrejón B, Borrero‐Palacios A, del Puerto‐Nevado L, Martínez‐Useros J, et al. Vitamin C uncouples the Warburg metabolic switch in KRAS mutant colon cancer. Oncotarget. 2016;7(30):47954–65.
Brandt KE, Falls KC, Schoenfeld JD, Rodman SN, Gu Z, Zhan F, et al. Augmentation of intracellular iron using iron sucrose enhances the toxicity of pharmacological ascorbate in colon cancer cells. Redox Biol. 2018;14(July 2017):82–7.
Cenigaonandia‐Campillo A, Serna‐Blasco R, Gómez‐Ocabo L, Solanes‐Casado S, Baños‐Herraiz N, Del Puerto‐Nevado L, et al. Vitamin C activates pyruvate dehydrogenase (PDH) targeting the mitochondrial tricarboxylic acid (TCA) cycle in hypoxic KRAS mutant colon cancer. Theranostics. 2021;11(8):3595–606.
Mamede AC, Pires AS, Abrantes AM, Tavares SD, Gonçalves AC, Casalta‐Lopes JE, et al. Cytotoxicity of ascorbic acid in a human colorectal adenocarcinoma cell line (WiDr): in vitro and in vivo studies. Nutr Cancer. 2012;64(7):1049–57.
Nakanishi K, Hiramoto K, Ooi K. High‐dose vitamin C exerts its anti‐cancer effects in a Xenograft model of Colon Cancer by suppressing angiogenesis. Biol Pharm Bull. 2021;44(6):884–7.
Pires AS, Marques CR, Encarnação JC, Abrantes AM, Mamede AC, Laranjo M, et al. Ascorbic acid and colon cancer: an oxidative stimulus to cell death depending on cell profile. Eur J Cell Biol. 2016;95(6–7):208–18.
Wang G, Yin T, Wang Y. In vitro and in vivo assessment of high‐dose vitamin C against murine tumors. Exp Ther Med. 2016;12(5):3058–62.
Yun J, Mullarky E, Lu C, Bosch KN, Kavalier A, Rivera K, et al. Vitamin C selectively kills KRAS and BRAF mutant colorectal cancer cells by targeting GAPDH. Science. 2015;350(6266):1391–6.
Nakanishi K, Hiramoto K, Sato EF, Ooi K. High‐dose vitamin C administration inhibits the invasion and proliferation of melanoma cells in mice ovary. Biol Pharm Bull. 2021;44(1):75–81.
Chen XY, Chen Y, Qu CJ, Pan ZH, Qin Y, Zhang X, et al. Vitamin C induces human melanoma A375 cell apoptosis via Bax‐ and Bcl‐2‐mediated mitochondrial pathways. Oncol Lett. 2019;18(4):3880–6.
Kang JS, Cho D, Kim Y‐I, Hahm E, Yang Y, Kim D, et al. L‐ascorbic acid (vitamin C) induces the apoptosis of B16 murine melanoma cells via a caspase‐8–Independent pathway. Cancer Immunol Immunother. 2003;52(11):693–8.
Mustafi S, Sant DW, Liu Z‐J, Wang G. Ascorbate induces apoptosis in melanoma cells by suppressing Clusterin expression. Sci Rep. 2017;7(1):3671.
Serrano OK, Parrow NL, Violet P‐C, Yang J, Zornjak J, Basseville A, et al. Antitumor effect of pharmacologic ascorbate in the B16 murine melanoma model. Free Radic Biol Med. 2015;87:193–203.
Du J, Martin SM, Levine M, Wagner BA, Buettner GR, Wang S, et al. Mechanisms of Ascorbate‐induced cytotoxicity in pancreatic Cancer. Clin Cancer Res. 2010;16(2):509–20.
Pollard HB, Levine MA, Eidelman O, Pollard M. Pharmacological ascorbic acid suppresses syngeneic tumor growth and metastases in hormone‐refractory prostate cancer. In Vivo. 2010;24(3):249–55.
Li Z, He P, Luo G, Shi X, Yuan G, Zhang B, et al. Increased Tumoral microenvironmental pH improves cytotoxic effect of pharmacologic ascorbic acid in castration‐resistant prostate Cancer cells. Front Pharmacol. 2020;11:570939.
Chen P, Yu J, Chalmers B, Drisko J, Yang J, Li B, et al. Pharmacological ascorbate induces cytotoxicity in prostate cancer cells through ATP depletion and induction of autophagy. Anti‐Cancer Drugs. 2012;23(4):437–44.
Ramezankhani B, Taha MF, Javeri A. Vitamin C counteracts miR‐302/367‐induced reprogramming of human breast cancer cells and restores their invasive and proliferative capacity. J Cell Physiol. 2019;234(3):2672–82.
Xu Y, Guo X, Wang G, Zhou C. Vitamin C inhibits metastasis of peritoneal tumors by preventing spheroid formation in ID8 murine epithelial peritoneal Cancer model. Front Pharmacol. 2020;11:645.
Gregoraszczuk EL, Zajda K, Tekla J, Respekta N, Zdybał P, Such A. Vitamin C supplementation had no side effect in non‐cancer, but had anticancer properties in ovarian cancer cells. Int J Vitam Nutr Res. 2020;3:1–11.
Lv H, Wang C, Fang T, Li T, Lv G, Han Q, et al. Vitamin C preferentially kills cancer stem cells in hepatocellular carcinoma via SVCT‐2. npj Precis Oncol. 2018;2(1):1.
Alyoussef A, Al‐Gayyar MMH. Cytotoxic and partial hepatoprotective activity of sodium ascorbate against hepatocellular carcinoma through inhibition of sulfatase‐2 in vivo and in vitro. Biomed Pharmacother. 2018;103:362–72.
Volta V, Ranzato E, Martinotti S, Gallo S, Russo MV, Mutti L, et al. Preclinical Demonstration of Synergistic Active Nutrients/Drug (AND) Combination as a Potential Treatment for Malignant Pleural Mesothelioma. PLoS One. 2013;8(3):e58051.
Ranzato E, Biffo S, Burlando B. Selective Ascorbate toxicity in malignant mesothelioma. Am J Respir Cell Mol Biol. 2011;44(1):108–17.
Su X, Shen Z, Yang Q, Sui F, Pu J, Ma J, et al. Vitamin C kills thyroid cancer cells through ROS‐dependent inhibition of MAPK/ERK and PI3K/AKT pathways via distinct mechanisms. Theranostics. 2019;9(15):4461–73.
Tronci L, Serreli G, Piras C, Frau DV, Dettori T, Deiana M, et al. Vitamin C cytotoxicity and its effects in redox homeostasis and energetic metabolism in papillary thyroid carcinoma cell lines. Antioxidants. 2021;10(5):809.
Zhou J, Chen C, Chen X, Fei Y, Jiang L, Wang G. Vitamin C promotes apoptosis and cell cycle arrest in Oral squamous cell carcinoma. Front Oncol. 2020;10:976.
Deubzer B, Mayer F, Kuçi Z, Niewisch M, Merkel G, Handgretinger R, et al. H2O2‐mediated cytotoxicity of pharmacologic Ascorbate concentrations to neuroblastoma cells: potential role of lactate and ferritin. Cell Physiol Biochem. 2010;25(6):767–74.
Castro M, Carson G, McConnell M, Herst P. High dose Ascorbate causes both Genotoxic and metabolic stress in Glioma cells. Antioxidants. 2017;6(3):58.
Campbell EJ, Dachs GU. Current limitations of murine models in oncology for Ascorbate research. Front Oncol. 2014;4:282.
Campbell EJ, Vissers MCM, Wohlrab C, Hicks KO, Strother RM, Bozonet SM, et al. Pharmacokinetic and anti‐cancer properties of high dose ascorbate in solid tumours of ascorbate‐dependent mice. Free Radic Biol Med. 2016;99:451–62.
Chen P, Stone J, Sullivan G, Drisko JA, Chen Q. Anti‐cancer effect of pharmacologic ascorbate and its interaction with supplementary parenteral glutathione in preclinical cancer models. Free Radic Biol Med. 2011;51(3):681–7.
Taper HS, Jamison JM, Gilloteaux J, Summers JL, Calderon PB. Inhibition of the development of metastases by dietary vitamin C:K 3 combination. Life Sci. 2004;75(8):955–67.
Alessandro Magrì et al: High-dose vitamin C enhances cancer immunotherapy: Sci Transl Med 2020 Feb 26;12(532):eaay8707. doi: 10.1126/scitranslmed.aay8707.
Franziska Böttger et al. High-dose intravenous vitamin C, a promising multi-targeting agent in the treatment of cancer: J Exp Clin Cancer Res 2021 Oct 30;40(1):343. doi: 10.1186/s13046-021-02134-y.
Manuela Giansanti et al: High-Dose Vitamin C: Preclinical Evidence for Tailoring Treatment in Cancer Patients. Cancers (Basel) 2021 Mar 20;13(6):1428. doi: 10.3390/cancers13061428.
References for IVC monotherapy in palliative care and quality of life (EOL)
Polireddy K, Dong R, Reed G, Yu J, Chen P, Williamson S, et al. High dose parenteral Ascorbate inhibited pancreatic Cancer growth and metastasis: mechanisms and a phase I/IIa study. Sci Rep. 2017;7(1):17188.
Takahashi H, Mizuno H, Yanagisawa A. High‐dose intravenous vitamin C improves quality of life in cancer patients. Pers Med Universe. 2012;1(1):49–53.
Vollbracht C, Schneider B, Leendert V, Weiss G, Auerbach L, Beuth J. Intravenous vitamin C administration improves quality of life in breast cancer patients during chemo−/radiotherapy and aftercare: results of a retrospective, multicentre, epidemiological cohort study in Germany. In Vivo. 2011;25(6):983–90.
Yeom CH, Jung GC, Song KJ. Changes of terminal Cancer patients’ health‐related quality of life after high dose vitamin C administration. J Korean Med Sci. 2007;22(1):7.
